- Clinical Technology
- Adult Immunization
- Hepatology
- Pediatric Immunization
- Screening
- Psychiatry
- Allergy
- Women's Health
- Cardiology
- Pediatrics
- Dermatology
- Endocrinology
- Pain Management
- Gastroenterology
- Infectious Disease
- Obesity Medicine
- Rheumatology
- Nephrology
- Neurology
- Pulmonology
Multiple Aorto-Cavitary Fistulas
Infective endocarditis (IE) starts as a vegetation on the valvular structures. The infection can extend to the adjacent periannular areas and erode into nearby cardiac chambers, leading to an aorto-cavitary fistula (ACF).1,2
Key words: Infective endocarditis, Aorto-cavitary fistula, Congestive heart failure
Infective endocarditis (IE) starts as a vegetation on the valvular structures. The infection can extend to the adjacent periannular areas and erode into nearby cardiac chambers, leading to an aorto-cavitary fistula (ACF).1,2 This can precipitate congestive heart failure (CHF) and hemodynamic instability secondary to shunting.1-3 In the setting of IE, only the aortic valve is affected. ACF is associated with high mortality even when surgical correction is performed. The incidence of ACF is about 1% up to 1.6%.1,3,4 In one series, the incidence of ACF in prosthetic valve endocarditis was 5.8%.5 More rare is the development of multiple ACFs, which is thought to occur in fewer than 0.5% of cases of endocarditis. We describe an adult with multiple ACFs who experienced decompensation despite intravenous antibiotic therapy and other medical interventions. He was successfully treated surgically.
Case report
A 51-year-old man with a history of intravenous drug use presented with a 7-day history of increasing dyspnea and a temperature reaching 39.5°C (103°F). Febrile episodes were associated with severe rigors, and the dyspnea had worsened during the 3 days before presentation. He was actively using intravenous heroin.
Physical examination demonstrated an acutely ill man with a temperature of 38.8°C (101.8°F). His blood pressure was 142/76 mm Hg with a pulse of 88 beats per minute, and his oxygen saturation was 96% on room air. Auscultation of the heart and lungs revealed bilateral rales and a grade 4/6 systolic murmur heard at the apex. The patient was unaware of ever being told that he had a heart murmur. In addition, significant lower extremity edema was observed.
Results of initial laboratory tests suggested leukocytosis. The white blood cell count was 13,100/µL with a left shift, and the serum creatinine level was 2.3 mg/dL. Chest radiographic findings were consistent with acute pulmonary edema and mild cardiomegaly, although an underlying infiltrate could not be excluded. Findings on his ECG were normal.
Treatment of IE was begun with intravenous vancomycin and gentamicin. Moxifloxacin was added for treatment of possible community-acquired pneumonia. Aggressive diuresis also was begun with intravenous furosemide every 12 hours.
Within 2 days of the start of therapy, the patient improved clinically, but leukocytosis and fever did not resolve. Multiple blood cultures grew methicillin-susceptible Staphylococcus aureus. The antibiotic regimen was switched to intravenous oxacillin.
The blood cultures remained positive for 4 days despite appropriate antibiotic therapy. A transthoracic echocardiogram (TTE) showed no valvular vegetations. A subsequent transesophageal echocardiogram (TEE) revealed no vegetations, an abscess in the region of the aortic annulus, an abnormal communication between the aorta and left atrium consistent with sinus of Valsalva fistula and aneurysm (Figure), and an additional communication between the right coronary cusp and right atrium.
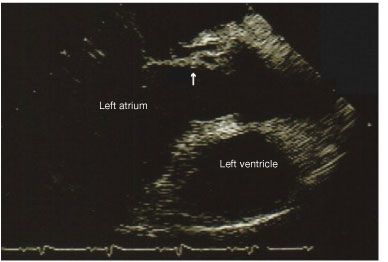
Figure –This echocardiogram shows abnormal communication between the aorta and left atrium, suggesting a sinus of Valsalva fistula and an aneurysm.
On the sixth hospital day, acute pulmonary edema developed. Inotropic and vasopressive agents were administered. Third-degree heart block developed, and the patient was taken immediately to the operating room for valve replacement.
Findings in the operating room included aortic valve vegetations, an annular abscess at the left coronary cusp with left ventricular to right atrial fistula and left ventricular to left atrial fistula. Aortic valve replacement was performed. The aortic annular abscess was repaired using a Dacron patch and reconstruction, and the left ventricle to left atrium and left ventricle to right atrium fistulas also were repaired. A pacemaker was placed for management of heart block.
Postoperatively, the patient’s course was unremarkable. His temperature, white blood cell count, and creatinine level returned to normal. He was treated with intravenous oxacillin for 4 weeks and was later discharged. At a follow-up examination, the patient still had an audible murmur.
Discussion
ACF is a rare but serious complication of aortic valve IE.1,2 5-7 An abscess or pseudoaneurysm near the sinus of Valsalva can rupture and lead to ACF. ACF can occur because of surgical trauma after placement of a prosthetic aortic valve, acute chest trauma, dissection of the aorta, and IE.1,7 Microbiologically, the infectious organisms involved in ACF are the same as those in IE, with Streptococcus species being the most common followed by Staphylococcus.1,2,8 The infective bacteria have no effect on prognosis. This is in contrast to cases of noncomplicated IE in which Staphylococcus species are associated with a higher mortality rate.2 All cases of ACF described in the literature have involved the aortic valve.
In this patient, the initial pathology started in the aortic valve and progressively spread to the aortic annulus. A localized abscess developed in this contained area. Secondary to extensive tissue destruction and necrosis, the infection spread along the contiguous tissue planes to involve the left ventricle and right atrium, causing fistulas. In a large review of 76 patients with ACF, an equal distribution of all 4 cardiac chambers involved in fistulous tracts was seen.1,5 Of these, 78% had annular abscesses. Of note, in our patient, the TTE did not reveal the fistulas later found on TEE. This is consistent with previous reports that show the TTE detected fistulas in 50% of cases whereas the TEE detected fistulas in 97% of cases.1,5,8
This patient followed a typical course seen in patients with ACF. Severe CHF because of intracardiac shunting rapidly developed several days after admission. In 2 recent reviews of patients with ACF, CHF developed in more than 60% of patients before undergoing surgery. The mean interval of diagnosis of ACF to surgery was 4.5 days.1,8 Eighty-seven percent of patients required surgical intervention and 92% of those patients required a valve replacement.1
Our patient’s heroin use was integral to the development of infection. He was maintained on a regimen of oral methadone while an inpatient. Methadone therapy continued after discharge, and the patient went on to undergo detoxification at a methadone clinic.
Although he eventually recovered from IE and its complications, mortality rates associated with ACFs have been as high as 41%, mostly due to multiorgan failure or septic or cardiogenic shock.1,3 Mortality rates are higher in those patients who require urgent surgical correction probably because they have complicating factors such as CHF and extensive anatomical destruction.2
Conclusion
Although rare, ACF is a serious complication of aortic valve IE that results in significant mortality despite appropriate medical or surgical therapy. Urgent transesophageal echocardiography is warranted to detect ACF and should strongly be considered in cases of aortic valve IE. Early diagnosis would facilitate prompt surgical intervention and provide information about prognosis.5
References:
REFERENCES
1. Anguera I, Miro JM, Vilacosta I, et al. Aorto-cavitary fistulous tract formation in infective endocarditis: clinical and echocardiographic features of 76 cases and risk factors for mortality. Eur Heart J. 2005;26:288-297.
2. Anguera I, Miro JM, Cabell CH, et al. Clinical characteristics and outcome of aortic endocarditis with periannular abscess in the International Collaboration on Endocarditis Merged Database. Am J Cardiol. 2005;96:976-981.
3. Anguera I, Quaglio G, Miro JM, et al. Aortocardiac fistulas complicating infective endocarditis. Am J Cardiol. 2001;87:652-654, A10.
4. Esen AM, Küçükoglu MS, Okçün B, et al. Transoesophageal echocardiographic diagnosis of aortico-left atrial fistula in aortic valve endocarditis. Eur J Echocardiogr. 2003;4:221-222.
5. Jenkins NP, Habib G, Prendergast BD. Aorto-cavitary fistulae in infective endocarditis: understanding a rare complication through collaboration. Eur Heart J. 2005;26:213-214.
6. Ananthasubramaniam K. Clinical and echocardiographic features of aorto-atrial fistulas. Cardiovasc Ultrasound. 2005;3:1.
7. Bayer AS, Bolger AF, Taubert KA, et al. Diagnosis and management of infective endocarditis and its complications. Circulation. 1998;98:2936-2948.
8. Anguera I, Miro JM, Evangelista A, et al. Periannular complications in infective endocarditis involving native aortic valves. Am J Cardiol. 2006;98:1254-1260.
