- Clinical Technology
- Adult Immunization
- Hepatology
- Pediatric Immunization
- Screening
- Psychiatry
- Allergy
- Women's Health
- Cardiology
- Pediatrics
- Dermatology
- Endocrinology
- Pain Management
- Gastroenterology
- Infectious Disease
- Obesity Medicine
- Rheumatology
- Nephrology
- Neurology
- Pulmonology
Fluticasone Exhalation Delivery System Reduced Exacerbations, Need for Antibiotics in Patients with Chronic Sinusitis
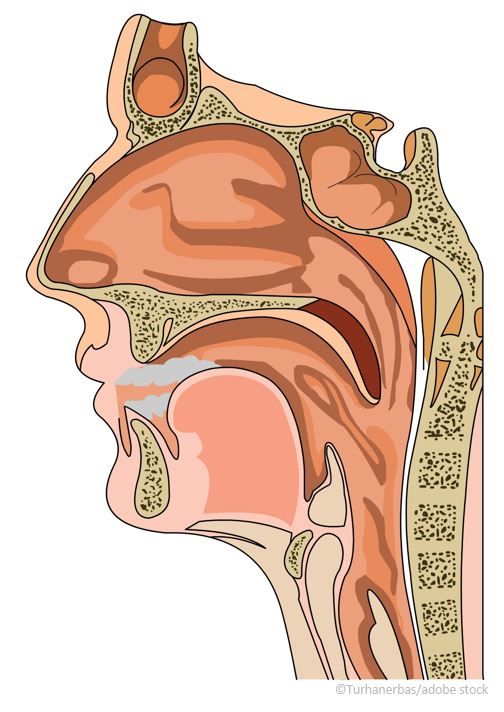
Use of a drug-device combination product delivering fluticasone propionate (XHANCE®, Optinose) nasal spray in patients with chronic sinusitis (CS) led to a significant decrease in acute exacerbations and need for antibiotics, according to new research presented during IDWeek 2022, held from October 19 to 23, in Washington, DC.
Findings from the landmark ReOpen phase 3 clinical trial program showed a greater than 60% decrease in acute exacerbations and need for antibiotics in patients with CS who used a drug-device combination product delivering fluticasone propionate nasal spray (also known as EDS-FLU), according to an Optinose press release.
Acute exacerbations of CS are associated with antibiotic and steroid use and for some persons surgery is required, according to Optinose. Moreover, chronic rhinosinusitis is responsible for 10 million outpatient visits annually and antibiotics are prescribed in approximately 70% of those visits. In fact, chronic rhinosinusitis is thought to be the number 1 reason for outpatient adult antibiotic prescriptions in the US, the company says.
The ReOpen clinical trial program is comprised of 2 global, randomized, double-blind, placebo-controlled trials that evaluated the efficacy and safety of EDS-FLU over 24 weeks, in patients with CS. ReOpen1 included 332 patients with CS with or without nasal polyps, and ReOpen2 included 222 patients with CS who did not have nasal polyps.
“Top-line results from these trials are the first ever that we are aware of that show improvement in both symptoms and inflammation inside the sinuses, and reduction in acute exacerbations of disease, with a nasal therapy for chronic sinusitis patients, including those with and without nasal polyps,” wrote the manufacturer in the release.
EDS-FLU received approval from the US Food and Drug Administration (FDA) in 2017 for the treatment of nasal polyps in patients aged ≥18 years. EDS-FLU is a drug-device combination of Optinose’s Exhalation Delivery System™ and fluticasone and has 2 features—the sealing nosepiece goes into a nostril and the mouthpiece is held in the mouth. The patient exhales through the mouthpiece, which forces the anti-inflammatory medication into the upper reaches of the nose more effectively than nasal spray.
If approved for CS, EDS-FLU would be the first drug to ever receive FDA approval for the treatment of CS either with or without nasal polyps, added the company.
Reference: Mahmoud R. Exhalation Delivery System with Fluticasone (EDS-FLU) Significantly Reduces Acute Exacerbations and Associated Antibiotic Use in Chronic Rhinosinusitis (LB1581). Poster presented at IDWeek 2022; October 21, 2022; Washington, DC
