- Clinical Technology
- Adult Immunization
- Hepatology
- Pediatric Immunization
- Screening
- Psychiatry
- Allergy
- Women's Health
- Cardiology
- Pediatrics
- Dermatology
- Endocrinology
- Pain Management
- Gastroenterology
- Infectious Disease
- Obesity Medicine
- Rheumatology
- Nephrology
- Neurology
- Pulmonology
Effect of Health Plan and Physician Clinic Collaboration on Increasing Omeprazole Use
A proposal for a pilot project to save money for a health plan was developed collaboratively with a clinic manager and the medical director of a local clinic. The goal was to encourage and support physicians in changing the proton pump inhibitor (PPI) medication prescribed for patients from a brand-name PPI to omeprazole, a more cost-effective generic option. The health plan identified members who had filled a prescription for a brand-name PPI and asked their physicians to consider switching the patients’ therapy to omeprazole. If the physician agreed to the change, the clinic would then send a letter to the patient, in which the physician recommended the change along with a new prescription for omeprazole. Following successful implementation in the initial pilot clinic, the program was extended to 4 more clinics. After achieving significant cost savings at all 5 clinics, the health plan is now expanding the program to more clinics as well as considering launching similar programs targeting other medication classes. (Drug Benefit Trends. 2009;21:158-163)
Proton pump inhibitors (PPIs) represent a costly therapeutic drug class for most health plans. PPIs are potent inhibitors of gastric acid secretion. Marketed PPIs include omeprazole (Prilosec), lansoprazole (Prevacid), rabeprazole (AcipHex), esomeprazole (Nexium), and pantoprazole (Protonix), of which only omeprazole and pantoprazole are currently available generically. These medications are used to treat persons with acid- related disorders, including peptic ulcer disease, gastroesophageal reflux disease, and Zollinger-Ellison syndrome; to treat and prevent gastroduodenal ulcers associated with the use of NSAIDs; and as part of a treatment regimen for the eradication of Helicobacter pylori.
The available PPIs are similar in chemical structure, mechanism of action, and effectiveness. Although some differences in effectiveness of PPIs have been reported, the magnitude of the differences has been small, with uncertain clinical importance.1-3 At the same time, the acquisition price of omeprazole has dropped to a point at which many health plans are implementing programs to encourage providers and members to favor the use of omeprazole over other PPIs. While the health plan evaluated strategies to increase its physicians’ generic prescribing ratio, the class of PPIs presented an opportunity to promote therapeutic substitution from brand-name PPIs to generically available omeprazole while maintaining similar clinical effectiveness.
Effective July 2007, SelectHealth requires step therapy using omeprazole before the plan will cover a different PPI. The other covered PPI options during the study period were lansoprazole and rabeprazole at a tier 2 (preferred brand) co-pay/coinsurance level. Esomeprazole was in tier 3 (nonpreferred brand) and was covered only if the patient’s condition did not improve following trials of omeprazole, lansoprazole, and rabeprazole. Pantoprazole became available as a generic during the study period; it was covered at tier 1 and also required step therapy. Switching to omeprazole is also encouraged through SelectHealth’s GenericSample program, under which members can try certain generic prescription drugs at no cost (on a one-time basis per medication) through participating retail pharmacies.
One difficulty for physicians is the identification of patients for whom therapeutic substitution that favors a generic may be applicable. With a recently instituted step-therapy requirement to try omeprazole first, patients naive to PPI therapy could receive a prescription for a brand-name PPI and take it to the pharmacy only to find out that it is not covered without a prior trial of omeprazole. However, patients who had previously filled a prescription for a brand-name PPI would be allowed to use up their remaining brand-name PPI refills. Another barrier is the difficulty in offering a therapeutic alternative to patients outside their routine office visits.
When the health plan, in collaboration with clinical management and physicians, launched a pilot program with 1 outpatient clinic, the share of omeprazole among all PPIs prescribed at this clinic was low compared with the health plan’s overall utilization of omeprazole. The health plan supplied the clinic with past PPI utilization data, and the clinic mailed out prescriptions for omeprazole. After evaluating the effects of collaboration with the pilot clinic, the program was then expanded to 4 other pilot clinics.
Methodology
In 2007, SelectHealth sought to increase the use of generically available omeprazole relative to other brand-name PPIs to reduce costs. This study evaluates changes in omeprazole utilization as a result of targeted efforts to encourage physicians to consider prescribing omeprazole in place of brand-name PPIs. One clinic (clinic A) was chosen as an initial pilot site for the intervention. The intervention was designed by the health plan in collaboration with a clinic manager and discussed with the medical director and lead physician at the clinic site. Utilization data were gathered for the 3-month period, or quarter, before the intervention, the quarter during the intervention, and the quarter following the intervention.
The study’s second phase measured omeprazole utilization at 4 additional clinics. For an overview of the methodology used, see Study Time Line. The study design was approved by the Intermountain Healthcare Office of Research and Institutional Review Board.
The health plan’s paid prescription claims were used to identify members who filled prescriptions for brand-name PPIs written by physicians at clinic A. Specifically, 245 members were identified who received a prescription for a brand-name PPI between April 1 and September 30, 2007. Member information was given to the clinic and subsequently evaluated by the physician who determined whether a new prescription for omeprazole would be appropriate in place of a brand-name PPI. The clinic created a letter in which the member’s physician described the opportunity for generic drug savings, and explained that all PPIs have a similar mechanism of action and clinical efficacy. Along with the letter, each targeted member received a prescription for omeprazole with 6 refills. The letters were sent in October 2007. Members could then decide whether they wanted to try omeprazole or continue their current therapy.
Prescription claims were analyzed 5 months after the letters were sent in order to capture data from the quarter before the intervention (2007Q3), the quarter during the intervention (2007Q4), and the quarter following the intervention (2008Q1). Claims were analyzed by the number of omeprazole prescriptions, PPI prescriptions, members taking omeprazole, and members taking any PPI. The market share of omeprazole prescriptions at clinic A was then compared with the health plan’s overall market share of omeprazole prescriptions, which was used as a benchmark. The health plan’s overall prescription share was calculated by dividing the number of the plan’s paid omeprazole claims by the number of all PPI paid claims.

After evaluating omeprazole utilization data from clinic A, the intervention was expanded to 4 more clinics (clinics B, C, D, and E). Members at these clinics were included in the study if they had filled a prescription for a brand-name PPI between April 1 and June 30, 2008. As with the original pilot clinic, prescribing physicians reviewed the list of members identified by the health plan’s claims data to determine which members should receive letters inviting them to switch to omeprazole. A medical assistant at each clinic administered the patient mailings and electronic medical record modifications. After the experience of clinic A, a template letter (Figure 1) was created and the process was streamlined to make the initiative more time-efficient for the other clinics.
Letters and prescriptions for omeprazole for the second intervention were sent in July and August 2008. Prescription claims were analyzed to compare preintervention and postintervention data and to compare utilization of omeprazole at clinics B, C, D, and E with the health plan’s overall utilization of omeprazole.
Results
The interventions resulted in increased numbers of prescriptions filled for omeprazole and a greater percentage of all PPI prescriptions filled for omeprazole. In the quarter before the intervention (2007Q3), market share of omeprazole was 24%, increasing to 39.5% in the quarter during the intervention (2007Q4) and to 48.2% in the quarter follow-ing the intervention (2008Q1). The health plan’s overall market share of omeprazole during the same time frames was 28.2% in 2007Q3, 34.3% in 2007Q4, and 39.7% in 2008Q1.
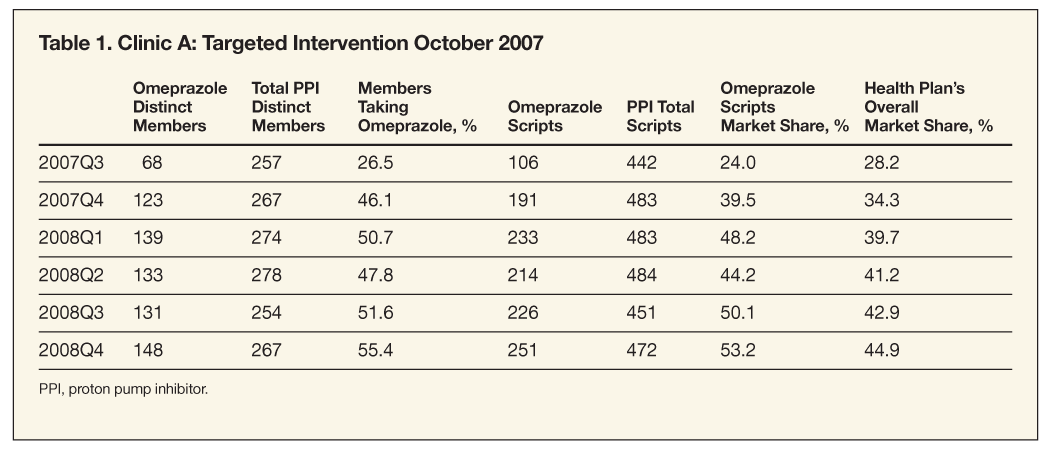
In 2007Q3, 68 members filled a prescription for omeprazole, representing 26.5% of all members who filled a prescription for a PPI during the quarter. The number of members who filled a prescription for omeprazole during 2007Q4 nearly doubled to 123, or 46.1% of members filling a prescription for a PPI. During 2008Q1, 139 members, or 50.7% of all members, receiving a PPI prescription received omeprazole. PPI utilization for clinic A and the health plan overall from mid-2007 through 2008 is shown in Table 1.
For the second intervention, a total of 453 members at clinics B, C, D, and E taking brand-name PPIs were eligible to receive letters and omeprazole prescriptions from their physicians. The clinics’ combined market share of omeprazole during the quarter before the intervention (2008Q2) was 34.8%, increasing to 39.5% for the quarter during the intervention (2008Q3), and 42.8% for the quarter after the intervention (2008Q4). The health plan’s overall market share of omeprazole during the same time frames was 41.2%, 42.9%, and 44.9%, respectively.
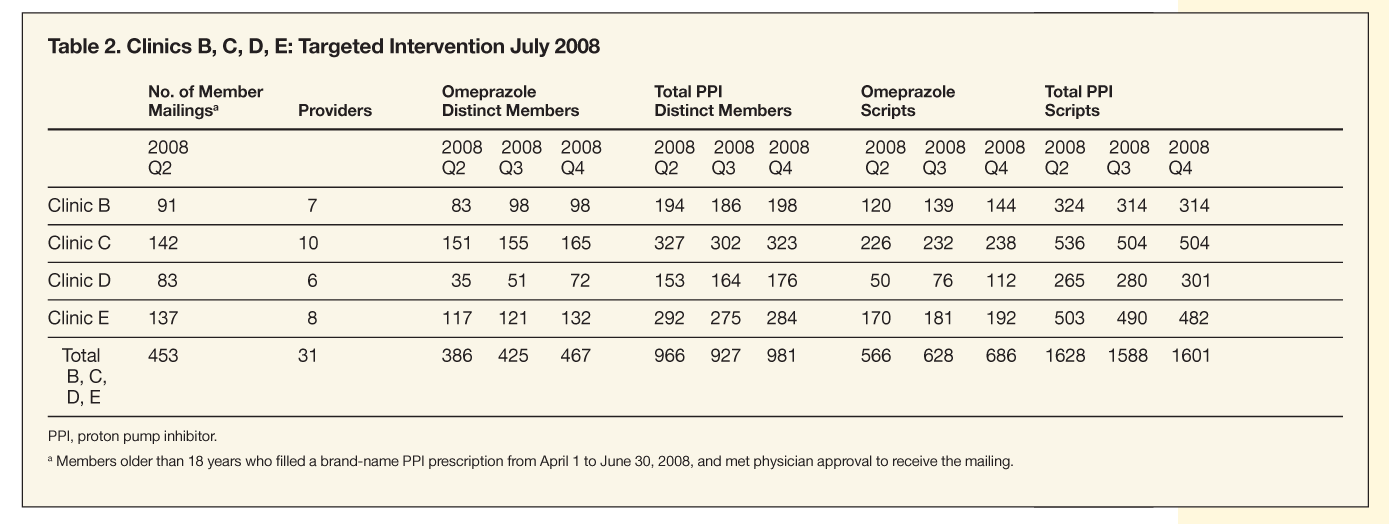
The percentage of members who filled prescriptions for omeprazole compared with that of all members who filled a prescription for any PPI was 40% for 2008Q2, 45.8% for 2008Q3, and 47.6% for 2008Q4. Individual clinic and combined omeprazole utilization, including number of members filling prescriptions for omeprazole and all PPIs for clinics B, C, D, and E, are shown in Table 2. Omeprazole market share for all 5 targeted clinics (A, B, C, D, and E) and the health plan’s overall market share of omeprazole are shown in Figure 2.

Drug cost avoidance has been estimated on the basis of the rate of omeprazole prescribing at the pilot clinics compared with the rate of omeprazole prescribing for the health plan’s entire book of business. Within the health plan, approximately 20% of brand-name and generic PPIs are filled as 90-day supplies. The daily average consumption of all PPI prescriptions is 1.22 doses per day. The average amount paid by the health plan (total amount less copay/coinsurance) per prescription is $37.13 for omeprazole compared with an average of $192.52 per prescription for the other PPIs.
For clinic A, market share of omeprazole increased by 108.8% (24% to 50.1%) from 2007Q3 to 2008Q3. During the same 1-year period, the market share of omeprazole for the health plan’s entire omeprazole utilization increased by 52.1% (28.2% to 42.9%). Multiplying the cost savings of $155.39 ($192.52 2 $37.13) by the number of switched prescriptions yields an estimated annual cost avoidance of $33,600 for clinic A.
Within 6 months, the omeprazole market share at clinics B, C, D, and E increased by 23% (34.8% to 42.8%) compared with 9% (41.2% to 44.9%) for the health plan’s entire omeprazole utilization. This results in an estimated annual cost avoidance of $43,700. The total annual cost avoidance for all 5 clinics is $77,300.
Discussion
This initiative was successful owing to a variety of factors. Having the support of the clinic manager and medical director was essential to complete the initiative in a timely manner and to emphasize its importance to prescribing physicians. Many physicians also viewed the initiative as a way to reach generic prescribing targets as well as an opportunity to help patients save on their copay/coinsurance amounts. From the members’ perspective, to receive a letter from their physician recommending a less costly therapeutic alternative probably has more impact than a similar letter from their health plan. In addition, to receive the prescription with the letter makes it easy and convenient for members to make the switch.
The calculated cost avoidance for this initiative is an estimate and will vary for different health plans on the basis of drug acquisition costs, pharmacy dispensing rates, negotiated rebates, and member cost-sharing formulas.
The market share of omeprazole within clinic A continued to increase at a greater overall rate than the health plan’s entire omeprazole utilization for a year after the initial intervention. On the basis of the performance of the initial pilot clinic, the second set of clinics may continue to increase their omeprazole market share compared with the health plan’s overall omeprazole utilization, resulting in greater cost avoidance than estimated during the next several quarters.
As seen in Table 1, there is a slight decrease in the market share of omeprazole from 48.2% in 2008Q1 to 44.2% in 2008Q2. In 2008Q1, 139 members filled 233 prescriptions for omeprazole, compared with 133 members filling 214 prescriptions in 2008Q2. The reason for the slight decrease in the number of members filling prescriptions for omeprazole and in the number of prescriptions for clinic A during this time frame is unknown. This reduction in market share from 2008Q1 to 2008Q2 is the only reduction in omeprazole market share during the 5 quarters observed for clinic A or the 3 quarters observed for clinics B, C, D, and E.
Limitations
All the pilot clinics that participated in the study had a market share of omeprazole that was lower than the health plan’s overall market share of omeprazole. This provides a greater opportunity for these clinics to rapidly increase their market share of omeprazole compared with clinics that are already prescribing at or above the benchmark, in this case the health plan’s overall market share of omeprazole. However, this is also one of the reasons these clinics were chosen.
A possible limitation to the expansion of this program is that clinics with an already high omeprazole market share may not benefit as much as clinics with a lower market share. This study did not include any interventions with clinics that had a high omeprazole market share, so the comparative effectiveness of the intervention is unknown. The maximum achievable omeprazole market share in an open, commercial formulary is also unknown.
Conclusion
Working in collaboration with physicians, the health plan increased the market share of omeprazole compared with other PPIs at selected clinics at a rate greater than the health plan’s overall market share of omeprazole. With the success seen in all 5 pilot clinics, the health plan is extending this program to more clinics in order to help physicians meet their generic prescribing goals and save money for both members and the health plan. The health plan is also considering implementing similar programs targeting other therapeutic classes to increase the use of generic alternatives.
Study Time Line
The health plan with the medical director and clinical management created a proposal at the initial pilot clinic (clinic A) in mid-2007.
Health plan claims were used to identify members who filled a prescription for a brand-name proton pump inhibitor (PPI) written by a physician from clinic A from April 1 to September 30, 2007.
Clinic A physicians reviewed their list of members for whom PPIs had been prescribed to determine whether omeprazole would be appropriate as a substitution for a brand-name PPI. In October 2007, clinic A sent to appropriate members a letter signed by their physician that emphasized an opportunity for savings through the use of generics and that included a prescription for omeprazole with 6 refills.
The health plan evaluated PPI claims from physicians at clinic A. Analysis included claims for the quarter before the intervention (2007Q3), claims for the quarter during the intervention (2007Q4), and claims for the quarter following the intervention (2008Q1).
After seeing the cost savings achieved by clinic A, the health plan expanded the pilot program to 4 additional clinics (clinics B, C, D, and E). Health plan claims were used to identify members who filled prescriptions for a brand-name PPI written by a physician at clinics B, C, D, and E from April 1 to June 30, 2008.
PPI claims from clinics B, C, D, and E were evaluated, including claims for the quarter before the intervention (2008Q2), claims for the quarter during the intervention (2008Q3), and claims for the quarter following the intervention (2008Q4).
References:
References
1. Ip S, Bonis P, Tatsioni A, et al. Comparative effectiveness of management strategies for gastroesophageal reflux disease. Agency for Healthcare Research and Quality. AHRQ publication 06-EHC003-EF. www.effectivehealthcare.ahrq. gov/repFiles/GERD%20Final%20Report.pdf. Published December 2005. Accessed April 3, 2009.
2. Wolfe MM. Overview and comparison of the proton pump inhibitors for the treatment of acid-related disorders. http://www.uptodate.com/ patients/content/topic.do?topicKey=~x/ 44UsYRYQJzHr. Updated February 9, 2009.
3. McDonagh MS, Carson S. Drug Class Review on Proton Pump Inhibitors. Portland, OR: Oregon Health and Science University; April 2004.
