- Clinical Technology
- Adult Immunization
- Hepatology
- Pediatric Immunization
- Screening
- Psychiatry
- Allergy
- Women's Health
- Cardiology
- Pediatrics
- Dermatology
- Endocrinology
- Pain Management
- Gastroenterology
- Infectious Disease
- Obesity Medicine
- Rheumatology
- Nephrology
- Neurology
- Pulmonology
A rare diagnosis: Endobronchial schwannoma
Endobronchial schwannomas are rare and often benign neoplasms that develop from the nerve sheath of the peripheral nervous system.
Endobronchial schwannomas are rare and often benign neoplasms that develop from the nerve sheath of the peripheral nervous system. We report a case of a solitary right-sided endobronchial schwannoma presenting with progressive respiratory symptoms. We discuss the clinical features of the disease, histopathological manifestations, diagnostic considerations, and therapeutic options.
The case
A 61-year-old man was referred to our pulmonary clinic for evaluation of dyspnea and wheezing. He complained of mild dyspnea at rest, wheezing in the supine position, and intermittent nonproductive nocturnal cough, progressing over the previous 4 months. He denied chest pain, orthopnea, paroxysmal nocturnal dyspnea, palpitations, hemoptysis, fever, chills, sweats, and gastroesophageal reflux.
The patient’s past medical history included hepatitis B and C, hypertension, dyslipidemia, obesity, obstructive sleep apnea, and depression. His surgical history included removal of an obstructing kidney stone 18 months earlier. His medications included inhaled fluticasone/salmeterol, inhaled levalbuterol, theophylline, montelukast, aspirin, atorvastatin, and atenolol. He had no known medication allergies. He denied tobacco use, excessive alcohol use, and illicit drug use.
His vital signs were normal except for an elevated blood pressure (163/93 mm Hg). Pulse oximetry showed an oxygen saturation of 97% on room air. Physical examination revealed a pleasant, obese man with diffuse expiratory wheezes, without rhonchi, rales, or dullness to percussion.
Pulmonary function testing revealed a forced expiratory volume in 1 second (FEV1) of 2.55 L (78% of predicted), forced vital capacity (FVC) of 3.59 L (82% of predicted), and FEV1:FVC ratio of 72%. Total lung capacity was mildly reduced at 4.87 L (75% of predicted), residual volume was reduced at 1.28 L (60% predicted), and slow vital capacity was reduced at 3.59 L (82% of predicted). His carbon monoxide–diffusing capacity was normal at 29.2 mL/min/mm Hg (108% of predicted). The flow-volume loop showed mild terminal coving of the expiratory limb, without evidence of inspiratory or expiratory blunting. The overall pattern was consistent with a mild restrictive ventilatory defect.
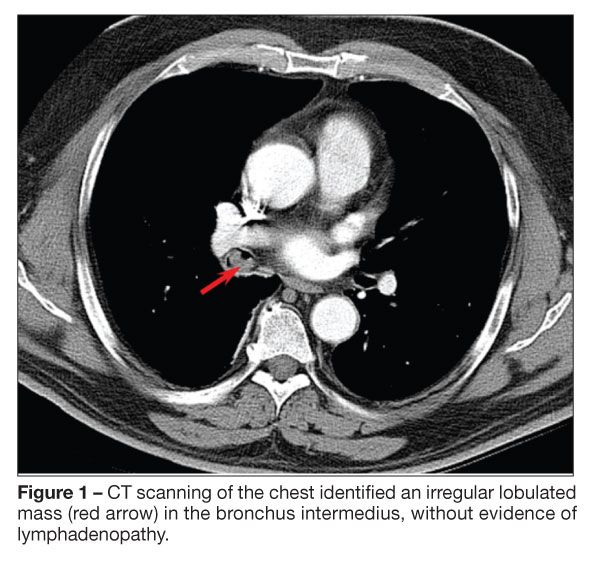
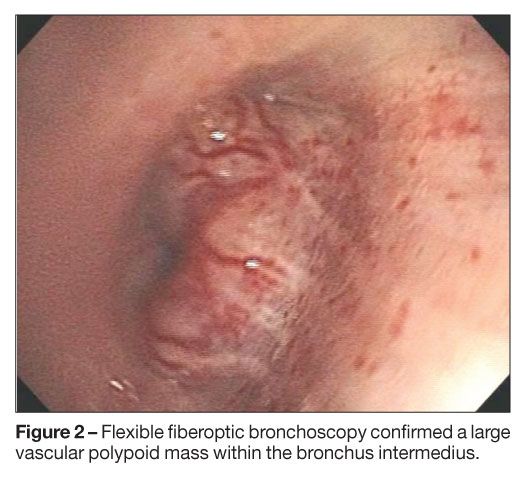
The initial chest radiographic findings suggested a possible superior segment, right lower lobe mass. A CT scan of the chest showed a 1-cm irregular lobulated mass in the bronchus intermedius without evidence of lymphadenopathy (Figure 1). Flexible fiberoptic bronchoscopy confirmed a large vascular polypoid mass within the bronchus intermedius (Figure 2). The bronchoscope was passed around the mass and beyond it into the distal airways, which appeared normal. Cytobrush and endobronchial biopsy specimens were taken. A small amount of bleeding occurred during the biopsy and stopped after application of topical phenylephrine (0.8 mg total).
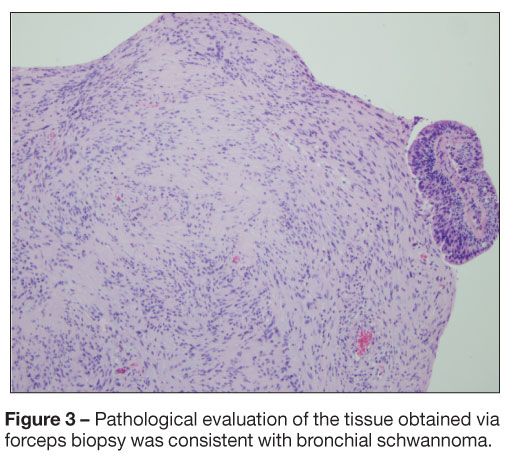
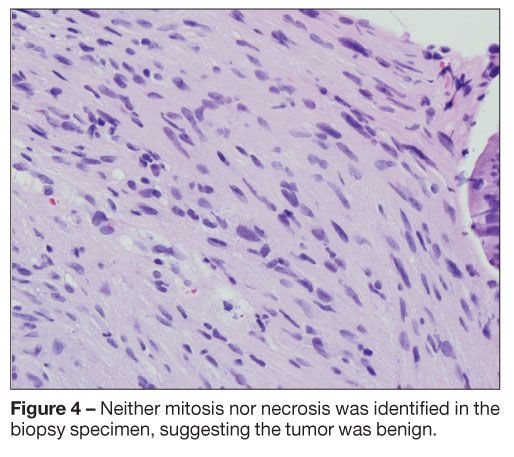
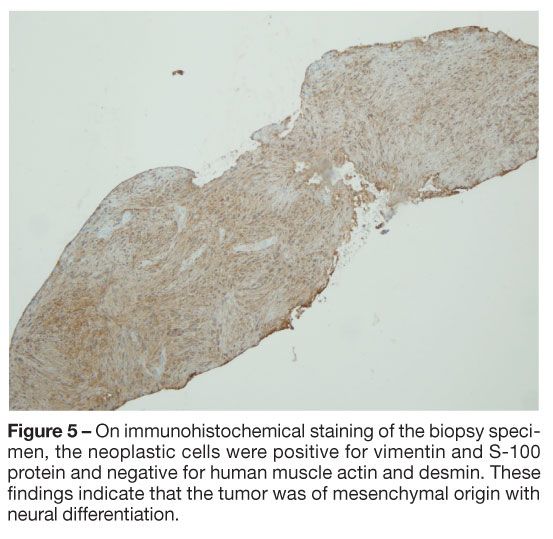
Pathological evaluation of the tissue obtained via forceps biopsy was consistent with bronchial schwannoma (Figure 3). Neither mitosis nor necrosis was identified, suggesting a benign tumor (Figure 4). On immunohistochemical staining, the neoplastic cells were positive for vimentin and S-100 protein and negative for human muscle actin and desmin, signifying that the tumor was of mesenchymal origin with neural differentiation (Figure 5). The cytobrush biopsy specimen did not reveal malignant cells.
After intensified treatment of gastroesophageal reflux disease and chronic bronchitis for 8 weeks, the patient reported less wheezing and cough and no dyspnea. Therapeutic options, including bronchoscopic laser treatment and surgical evaluation for tumor resection, were discussed with the patient. He decided to forego consultation with our surgical colleagues and opted for watchful waiting. Six months after diagnosis, a repeated chest CT scan revealed interval development of mild right lower lobe atelectasis, despite improved symptoms and stable tumor size of 1 cm.
Discussion
Endobronchial tumors can be either malignant or benign (Table 1).1-3 Neurogenic tumors are rare and can manifest at any age (with most occurring in young adulthood, after age 20 years).2 This type of tumor arises from peripheral nerves located within the bronchial wall, composed of nerve sheath Schwann cells, described as neurilemomas (schwannomas) or neurofibromas.4 Endobronchial schwannomas are extremely rare; the exact incidence is not known, although one institution reported that 0.2% of all lung tumors were primary neurogenic tumors.5,6
Schwannomas are benign tumors, are rarely associated with von Recklinghausen neurofibromatosis, and rarely undergo malignant transformation.2,4,7,8 Schwannomas can occur in all regions of the tracheobronchial tree, with intraluminal and/or extraluminal extensions.9-11 They can occur centrally in the airways as well as peripherally in the lung parenchyma.9 It is not clear whether central or peripheral lesions account for most of these tumors, but both schwannomas and neurofibromas can occur in the mediastinum, lung parenchyma, or tracheobronchial tree.4,7
Schwannomas are encapsulated, single tumors attached to a nerve that originates from the peripheral nervous system. Neurofibromas are not encapsulated, are multiple in number, and occur within the nerve sheath.4 Benign features of schwannomas include an encapsulated mass, rare or no mitotic figures, and borderline pleomorphism well demarcated from surrounding tissue.5 Malignant features include an unencapsulated mass, perineural invasion, indistinct cellular borders, frequent mitotic figures, pleomorphic fusiform cells, areas of hemorrhagic and cystic degeneration, and metastasis.5,6,12 Histologically, schwannoma spindle cells are arranged in a highly cellular and palisading nuclei pattern (Antoni A) or a less cellular and more fibrous pattern (Antoni B).3,13

Evaluation of respiratory symptoms, such as wheezing, stridor, dyspnea, cough, hemoptysis, and chest pain, usually prompts thoracic imaging.7,14 Chest radiographic findings may be unrevealing or normal, requiring more detailed imaging.1,2 CT and MRI scans can help determine the size of the mass and its relationship to adjacent tissue.2,4 Ventilation-perfusion scans have been used to evaluate perfusion to pulmonary regions of interest.6 Bronchoscopy is warranted if suspicious airway lesions are visualized on these imaging studies or if clinical suspicion is high. Evaluation by flexible fiberoptic bronchoscopy is more common than rigid bronchoscopy, but the latter has been reported.3,14
Biopsy specimens may be obtained during flexible bronchoscopy. If the neurogenic tumor is hypervascular, bronchoscopic biopsies should be performed in centers with experienced staff available to manage serious hemorrhagic complications.2 Interventional bronchoscopy and thoracic surgical expertise should be readily available. There are reports of surgical tumor resection without prior biopsy in patients with hypervascular tumors that are deemed too high a bleeding risk.4 Pathological evaluation of biopsy specimens should include immunohistochemical stains for vimentin and S-100 protein, to confirm neurogenic origin.
There is no uniform approach to the treatment of endobronchial schwannomas (Table 2). Historically, the standard treatment has been a surgical resection; however, given the benign nature of this tumor, less invasive options have evolved.15,16 Definitive treatment by tumor resection or laser therapy is recommended for symptomatic mass lesions not responsive to conservative management, such as bronchodilators and antibiotics, when indicated. However, the available therapeutic approaches are varied and must be carefully tailored to each specific clinical scenario.3,4,9,15,17 Watchful waiting is also an option for asymptomatic benign schwannomas in select patients.
The prognosis is generally good for benign lesions, as is expected in this case. Long-term follow-up may be needed given the slow growth of these tumors and the potential for recurrence after resection.2
References:
REFERENCES
1. Takeda S, Hashimoto T, Kusu T, Kawamura T, et al. Management and surgical resection for tracheobronchial tumors: institutional experience with 12 patients. Interact Cardiovasc Thorac Surg. 2007;6:484-489.
2. Righini CA, Lequeux T, Laverierre MH, Reyt E. Primary tracheal schwannoma: one case report and a literature review. Eur Arch Otorhinolaryngol. 2005;262:157-160.
3. Weiner DJ, Weatherly RA, DiPietro MA, Sanders GM. Tracheal schwannoma presenting as status asthmaticus in a sixteen-year-old boy: airway considerations and removal with the CO2 laser. Pediatr Pulmonol. 1998;25:393-397.
4. Hsu HS, Wang CY, Li WY, Huang MH. Endotracheobronchial neurofibromas. Ann Thorac Surg. 2002;74:1704-1706.
5. Roviaro G, Montorsi M, Varoli F, et al. Primary pulmonary tumours of neurogenic origin. Thorax. 1983;38:942-945.
6. Feldhaus RJ, Anene C, Bogard P. A rare endobronchial neurilemmoma (schwannoma). Chest. 1989;95:461-462.
7. Chen SR, Chen MH, Ho DM, et al. Massive hemoptysis caused by endobronchial schwannoma in a patient with neurofibromatosis 2. Am J Med Sci. 2003;325:299-302.
8. Rowlands D, Edwards C, Collins F. Malignant melanotic schwannoma of the bronchus. J Clin Pathol. 1987;40:1449-1455.
9. Kasahara K, Fukuoka K, Konishi M, et al. Two cases of endobronchial neurilemmoma and review of the literature in Japan. Intern Med. 2003;42:1215-1218.
10. Stack PS, Steckler RM. Tracheal neurilemmoma: case report and review of the literature. Head Neck. 1990;12:436-439.
11. Shirakusa T, Takada S, Yamazaki S, et al. Intrabronchial neurilemmoma-review of cases in Japan. Thorac Cardiovasc Surg. 1989;37:388-390.
12. McCluggage WG, Bharucha H. Primary pulmonary tumours of nerve sheath origin. Histopathology. 1995;26:247-254.
13. Shigematsu H, Aoe M, Date H. Schwannoma occurring from the lingular bronchus. Eur J Cardiothorac Surg, 2007;32:537.
14. Erol MM, Uzun H, Tekinbas C, et al. A case of intratracheal schwannoma presenting at the emergency department with a diagnosis of asthmatic attack. J Emerg Med. 2008 Apr 23; [Epub ahead of print].
15. Mizobuchi T, Iizasa T, Iyoda A, et al. A strategy of sequential therapy with a bronchoscopic excision and thoracotomy for intra- and extrabronchial wall schwannoma: report of a case. Surg Today. 2005;35:778-781.
16. Rusch VW, Schmidt RA. Tracheal schwannoma: management by endoscopic laser resection. Thorax. 1994;49:85-86.
17. Tsukada H, Osada H, Kojima K, Yamate N. Bronchial wall schwannoma removed by sleeve resection of the right stem bronchus without lung resection. J Cardiovasc Surg (Torino). 1998;39:511-513.
